Figure 5 from Dr. NO and Mr. Toxic – the versatile role of nitric oxide
4.5 (459) · € 20.99 · Auf Lager
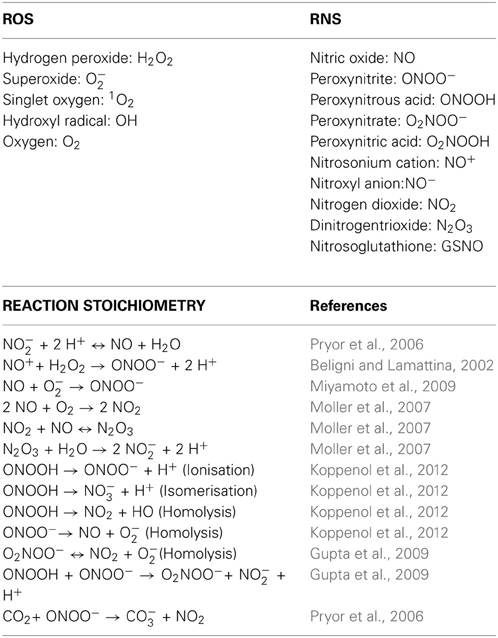
Figure 5: Reactive nitrogen species (RNS) formation: interaction of NO, superoxide, peroxynitrite and nitrogen dioxide. When NO and superoxide are present, they can react to generate peroxynitrite that is highly reactive. On the other hand, NO, superoxide and nitrogen dioxide (NO2) form dinitrogen trioxide (N2O3) and peroxynitrate (NO4−). Peroxynitrate breaks down to give nitrite and oxygen. Altogether, they are powerful nitrative and oxidative agents that can act on proteins, lipids and nucleic acids. - "Dr. NO and Mr. Toxic – the versatile role of nitric oxide"

IJMS, Free Full-Text

Dr. NO and Mr. Toxic – the versatile role of nitric oxide

Opportunities for Nitric Oxide in Potentiating Cancer

Nitric Oxide: Physiological Functions, Delivery, and Biomedical

Extracellular vesicles from differentiated stem cells contain

IJMS, Free Full-Text

Nitrification and beyond: metabolic versatility of ammonia

Dr. NO and Mr. Toxic – the versatile role of nitric oxide

Dr. NO and Mr. Toxic – the versatile role of nitric oxide